Request a Trial
Hoodin is specifically designed to perform monitoring for a life science product throughout their entire life cycle. With Hoodin, you have the flexibility to set up monitoring for a specific product (brand name) or a product type (such as a device, IVD product, or drug.) The trial project you will have access to will be customized to monitor the product or product type of your preference.
You will get access to a 14- day free trial of Hoodin
Request a Trial
Hoodin is specifically designed to perform monitoring for a life science product throughout their entire life cycle. With Hoodin, you have the flexibility to set up monitoring for a specific product (brand name) or a product type (such as a device, IVD product, or drug.) The trial project you will have access to will be customized to monitor the product or product type of your preference.
You will get access to a 14- day free trial of Hoodin
Request a Trial
Hoodin is specifically designed to perform monitoring for a life science product throughout their entire life cycle. With Hoodin, you have the flexibility to set up monitoring for a specific product (brand name) or a product type (such as a device, IVD product, or drug.) The trial project you will have access to will be customized to monitor the product or product type of your preference.
You will get access to a 14- day free trial of Hoodin
360 Surveillance &Vigilance
For Software as a Medical Device (SaMD)
Is your Software as a Medical Device (SaMD) company navigating the intricate landscape of regulations, standards, post-market surveillance, cyber security, privacy policies, and data-related regulations and laws? Look no further than Hoodin.
1
Comprehensive SaMD Compliance
Hoodin simplifies SaMD compliance with regulatory requirements and standards, ensuring that your company operates seamlessly within the complex regulatory framework.
Vigilant Post-Market Surveillance
Our robust monitoring tools keep you ahead of the curve by tracking adverse events and ensuring timely and appropriate responses, a critical aspect of SaMD compliance.
2
3
Cybersecurity at the Core
Hoodin keeps you informed about the latest changes, directives, and updates regarding cybersecurity, privacy policies, and data-related regulations and laws, allowing you to adapt seamlessly
Competitive Edge
Gain a competitive advantage with Hoodin's market monitoring tools, providing valuable insights into your industry landscape, and helping you adapt to changes in real-time, no matter where you are in the product lifecycle.
4
5
Continuous Regulatory Confidence
Trust Hoodin to keep you informed about evolving regulations, policies, and standards. Stay compliant at every stage with Hoodin's versatile support for your compliance needs
Experience 360 Surveillance & Vigilance!
Unlock the future of Medtech monitoring with Hoodin – where simplicity meets precision.
Effortless Trial
In just 2-3 hours, grasp the power of Hoodin. Our AI engines will craft a personalised Hoodin project, tailored to your unique medical device. Experience Hoodin today for 360 Surveillance & Vigilance.

High Value, Low Effort
The Hoodin platform ensures maximum value with minimal effort.
After your 14-day trial, you can choose to meet our experts for a detailed evaluation and validation of your tailored Hoodin project.

Experience Hoodin today – your gateway to streamlined, efficient SaMD monitoring with 360 Surveillance & Vigilance.
Key features
and unique values
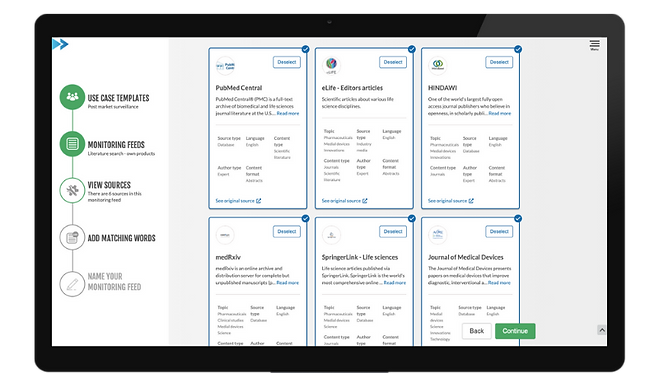
Monitors the most important and relevant sources for SaMD
Monitor both own and competitor products.
Export result and insight for use in reports.
Share results and insights with invited peers (free feature).
Keep track of all changes made in your Hoodin project with the included change-log feature.
Validated by industry experts.